Last week we reviewed viral COVID-19 tests and their accuracy in detecting SARS-CoV-2 virus in humans. This week we will discuss the other main type of testing: antibody tests.
Review and Recap of Viral Testing
People who symptoms consistent with the COVID-19 clinical case definition will have viral RNA, which can be detected using an RT-PCR test as early as 1 day into experiencing symptoms. These detectable levels of virus in the body decline over time, and rarely are found after 6 weeks, as you can see from this figure from last week’s post from JAMA: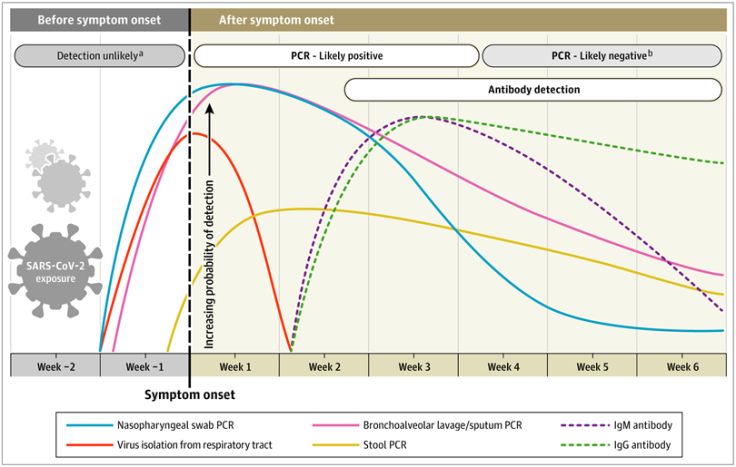 This chart shows us the limits of using viral detection tests for monitoring the spread and intensity of COVID-19 disease, because viral detection tests cannot easily tell us who has been infected in the course of the outbreak, nor are they likely to identify people who were exposed/infected and never had symptoms. So what testing can we use when current infection no longer exists?
This chart shows us the limits of using viral detection tests for monitoring the spread and intensity of COVID-19 disease, because viral detection tests cannot easily tell us who has been infected in the course of the outbreak, nor are they likely to identify people who were exposed/infected and never had symptoms. So what testing can we use when current infection no longer exists?
Antibody tests may be a solution
One solution is to look for and measure the immune response our bodies mount during and following infection with SARS-CoV-2. This is the second part to a two-pronged testing approach to controlling the pandemic, and the complement to viral detection tests.
Antibody testing allows us to identify evidence of prior infection through the detection of antibodies in blood. Antibody testing can be used for population surveillance, by helping to determine the prevalence of COVID-19 among (1) those known to have recovered from infection and (2) those who were unknowingly exposed/infected.
But, as much as antibody testing is making headlines as the silver-bullet to all our gaps in knowledge about who is and who is not vulnerable to continued spread of COVID-19, many questions remain about the utility and appropriateness of using antibody testing, and whether or not antibody tests can serve as “immunity passports”, to enable individuals to return to work and travel. We tackle these questions below and give you our take on the future of antibody testing for effectively controlling COVID-19 (spoiler alert: antibody tests are not a cure-all!).
What are antibodies?
Antibodies, also known as immunoglobulins, are proteins found in blood plasma that are produced and used by the immune system.
There are two main types of immunoglobulins: IgM and IgG:
- IgM antibodies are the first type of antibody made in response to an infection. IgM levels rise rapidly after your body is exposed to a new pathogen, and IgM levels begin to drop as IgG antibodies are produced for longer term protection.
- IgG antibodies are the most common antibody and provide longer-term immunity against pathogens, remaining in your blood for longer after the infection has passed.
When we get sick from an infection like COVID-19, healthy immune systems are designed to mount an attack on infected cells, producing neutralizing antibodies which can last in the body and diminish or eliminate the likelihood of re-infection from the same pathogen.
A recent review from Nature describes the chronology of events following SARS-CoV-2 infection:
Neutralizing antibodies disarm a virus’ ability to invade healthy cells, replicate and propagate in the body. We expect that these antibodies would do the same with future exposures to the virus. However, the durability of these antibodies over time is not the same for all pathogens. Some viral and bacterial infections lead to life-long pathogen-specific protection against future infections (like measles or smallpox viruses), but others we can get again and again (for example, gonorrhea).
Unfortunately, we do not yet know whether SARS-CoV-2 infection results in life-long immunity – or lasting immunity at all. We can look to other coronaviruses (like SARS-CoV-1) for clues about potential immunity. For SARS infection, individuals who developed neutralizing antibodies did have some levels of protection against re-infection, which lasted up to 2-3 years. However, only more research – and more time to see how antibody levels change in those who have recovered – will determine what sort of immunity exists to COVID-19 and how long we can expect that immunity to last. Through antibody testing, we can start to understand the potential levels of immunity in the population and seek to understand the duration of this potential protection over time.
What antibody testing is available and how do we interpret the test results?
Antibody tests determine the presence of IgM and/or IgG in blood specimen samples. There are currently > 100 antibody tests available in the US with widely variable quality. A recent study measured four commercial SARS-CoV-2 antibody tests and found that sensitivities ranged from 77.3% to 100%, and specificity ranged from 88.7% to 100%. (the estimates of sensitivity and specificity for select FDA-reviewed tests can be found on the FDA website). However, many of these tests can only indicate the presence or absence of COVID-19 antibodies, and cannot speak to what type of antibody is present – or if that antibody is indicates that there is immunity to future infection.
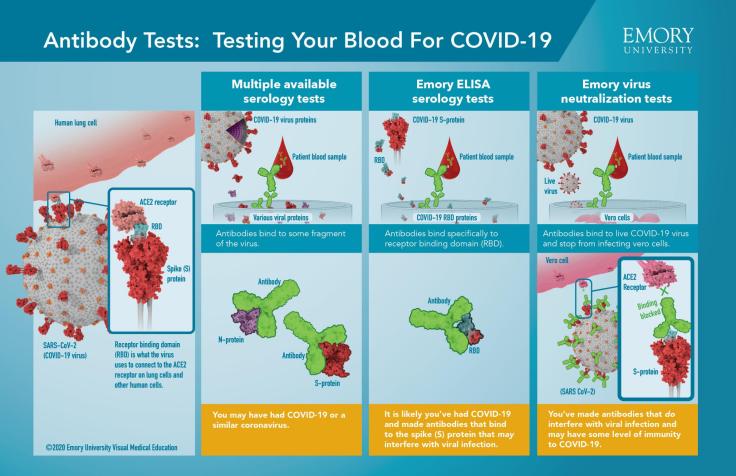 This useful infographic from Emory University can differentiate between these different types of serology tests and how to interpret their results. As the figure describes, some available serology tests (left-most panel), generate a positive result if the antibody binds to a fragment of the COVID-19 virus. It is possible that antibodies in the patient’s blood would trigger a positive test result if they have had COVID-19 infection or a similar coronavirus, because the binding is not very specific.
This useful infographic from Emory University can differentiate between these different types of serology tests and how to interpret their results. As the figure describes, some available serology tests (left-most panel), generate a positive result if the antibody binds to a fragment of the COVID-19 virus. It is possible that antibodies in the patient’s blood would trigger a positive test result if they have had COVID-19 infection or a similar coronavirus, because the binding is not very specific.
By contrast, the middle panel of tests use a COVID-19 specific receptor-binding domain. Here, a patient’s blood will generate a positive result if they have antibodies that can bind to COVID-19 spike protein, which is more specific to determining COVID-19 infection (i.e. – not any other coronavirus). However – neither of these two tests indicates anything about whether the patient is immune!
The last panel, the neutralization test, requires antibodies to bind to live virus and show their ability to block virus from infecting cells – this indicates that the antibodies in the patient’s blood are able to interrupt or completely halt the life-cycle of a viral infection, and may provide some level of immunity.
It’s important to recall that if you go to get antibody testing, you will likely be receiving a nonspecific test, so you might receive a test that can give false positives for other types of coronavirus infection.
Should you get an antibody test?
The discussion above about antibody tests being used to inform public health decision-making hinges upon one major assumption: The tests work. On May 27th, the CDC released new guidance that the available tests on the market might not be accurate enough to depend upon for making these sorts of public health decisions: specifically whether large groups can safely gather in schools, dorms, or correctional facilities. Why is this? Let’s get back to the basics of test accuracy in low-prevalence settings.
Negative and positive predictive values
The main reason that these tests are so hard to interpret has to do with the positive predictive value (PPV) and negative predictive value (NPV) of a test: the probability that individuals with positive (and negative) test results are truly antibody positive (and negative). The PPV and NPV are derived from the test sensitivity and specificity discussed last week – however they also depend on the percentage of individuals in the population who are truly antibody positive. As a reminder, the higher the sensitivity, the fewer false negatives a test will give. The higher the specificity, the fewer false positives.
When the prevalence is high (many individuals are truly antibody positive for COVID-19), the PPV will increase: this means that more people who test positive are truly positive. On the flip side, when the test is conducted in a population with low prevalence of antibodies for COVID-19, the PPV drops and the false positive rate increases. Across most of the country, even in the most hard hit areas, the prevalence of antibodies to SARS-CoV-2 is likely <5-25%.
The CDC explains that in a population where the prevalence is 5%, a test with 90% sensitivity and 95% specificity will have a PPV of 49% (For reference, starting May 4, this is the threshold for test accuracy that must be met for the FDA to issue an emergency use authorization). That means LESS THAN HALF of those who test positive truly have antibodies to COVID-19. However, if the same population has an antibody prevalence of 52%, the PPV would be > 95%. Here’s a nice simulation to understand why this happens from qz.com.

For the time being, CDC and many in the public health community are urging that antibody testing be used for surveillance purposes to monitor the spread and intensity of COVID-19. Unlike relying only on testing in people who seek medical care, wide-reaching serology-surveys (sero-surveys) can provide a snapshot at a specific point in time of how many people have been infected (and capture asymptomatic people who unknowingly were infected). Repeat sero-surveys in the same populations will help determine how long antibodies last, giving clues to possible immunity. This type of information may eventually help inform public health measures, such as social distancing and targeted vaccination, and planning for healthcare needs. Further information about the use of sero-surveys can be found here.
The Takeaway:
Serology tests are still being developed and evaluated. Improved regulation of these tests is a must. All told, the current prevalence of antibodies is low enough that a positive antibody result does not necessarily indicate the presence of COVID-19, due to high false positive test rates. Even with a positive test, we do not know that the presence of antibodies (or at what level of presence) signals protection against reinfection! False positive serology tests could cause false reassurance that one is no longer susceptible to COVID-19, resulting in a reduction in social distancing, which could spread the disease. As a result, serology testing for individual decision making is not yet useful. More accurate tests and better knowledge of what these tests mean in terms of immunity will help to make antibody tests useful for the next phase of managing the COVID-19 pandemic.
This week, my lab mate, Cara Janusz, a fellow PhD candidate in Epidemiology at the University of Michigan, joins with editing contributions to the blog. She is excited to support evidence synthesis and communication about our immunization research and policy interests on this platform! She is passionate about supporting evidence-based immunization policy and practice through her research.
Leave a comment